|
1/18/2024 0 Comments Cobalt electron configurationelectron configuration for cobalt is 1s2 2s2 2p6 3s2 3p6 3d7 4s2 Fort de. And in the case of cobalt, there are 8 valence electrons. View our Cobalt Boats a RamboMarine Cobalt 25SC Review 2020 23 1 New Cobalt CS23. Cobalt is in the first row of the d-block of elements. This electron configuration has the most unpaired electrons possible and is known as a high spin arrangement. The electron configuration of cobalt is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 7 4s 2. Valence electrons are e – (electrons) of the atom present in the outermost shell. Answer and Explanation: Become a member to unlock this answer Create your account. What is The Electron Configuration of CobaltĮlectron distribution of cobalt in s, p, d blocks of 4 orbits is 2, 8, 15, 2 and it can be written as: 1s 22s 22p 63s 23p 63d 74s 2. 5 BE) Our free-electron configuration calculator also depicts an abbreviated way of finding electron configuration. Before drawing the orbital diagram, you should know the three general rules. Now in the next step, start drawing the orbital diagram for cobalt. There are 2, 8, 15, 2 elements present in the 4 orbits of cobalt and can be represented in this form: 1s 22s 22p 63s 23p 63d 74s 2. Cobalt atoms have 27 electrons and the electronic shell structure is 2, 8, 15, 2 with Atomic Term Symbol (Quantum Numbers) 4F9/2. The electron configuration of cobalt is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 7. 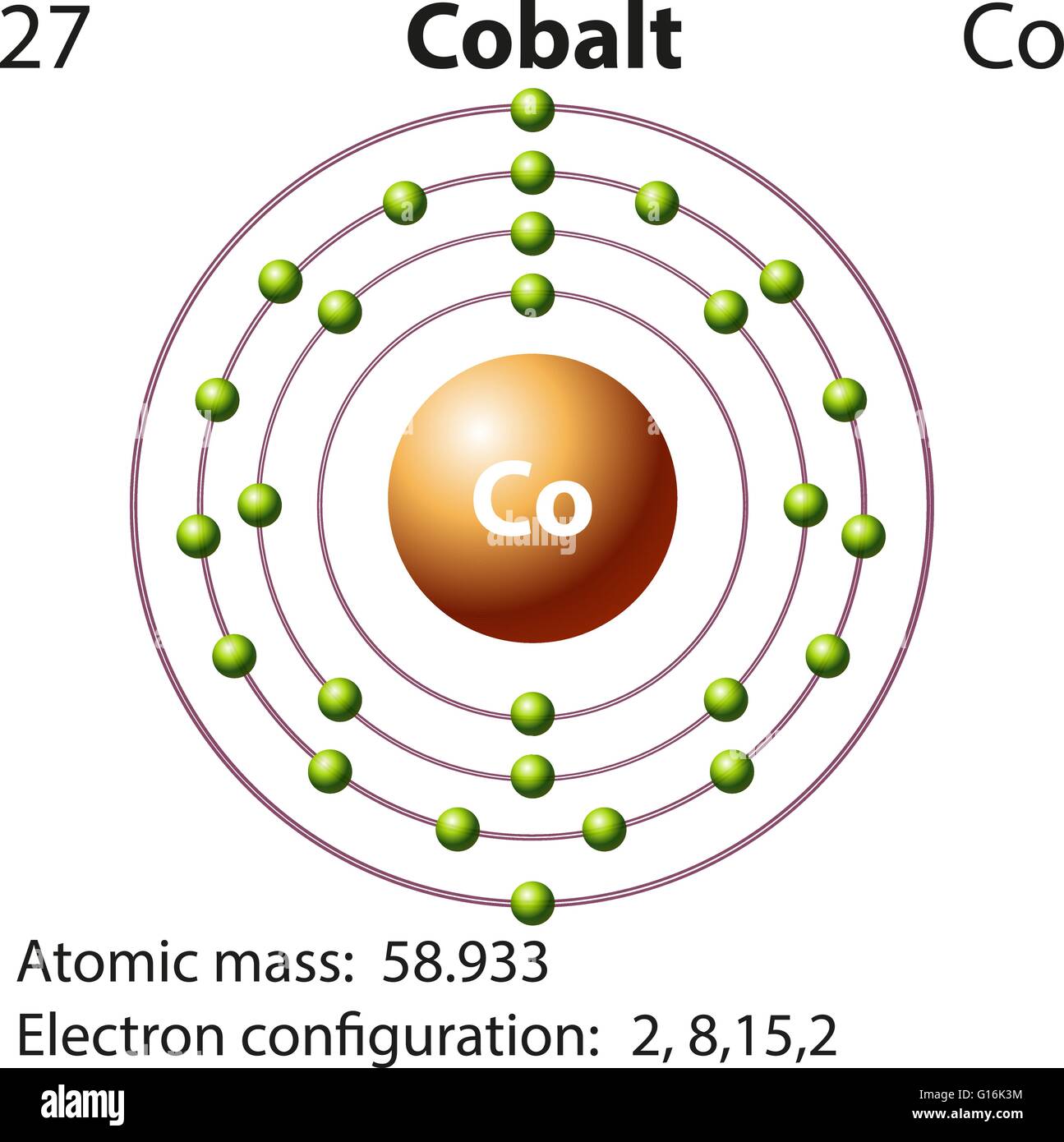
The valence electrons are leftover electrons that don’t fill a shell or satisfy the octet rule (except for noble gases) or 18. A noble gas core is the noble gas element symbol enclosed in brackets: He, Ne, Ar, Kr, Xe, or Rn. It further has a number of other significances in the domain of chemistry and quantum physics. The electron configuration for M2+ is Ar3d7, which means that the ground state electron configuration is Ar3d74s2, because the electrons are first. Write the noble gas configuration by writing the noble gas core, followed by the valence electrons. This electron configuration of the element represents the actual and the unique identity of the element. Electron configuration of Cobalt is Ar 3d7 4s2. Electron Configuration and Oxidation States of Cobalt. 
See the electron configuration diagram, the position of cobalt in the periodic table, and the rules for each subshell. To write the configuration for the Cobalt ions, first we need to write the electron configuration for just Cobalt (Co). Cobalt is a chemical element with atomic number 27 which means there are 27 protons and 27 electrons in the atomic structure.The chemical symbol for Cobalt is Co. \) b.Electron distribution of electrons in atom or molecules is defined as electron configuration and electron configuration for cobalt ion can be represented as:ġs 22s 22p 63s 23p 63d 74s 2. Or 3d 74s 2.įull electron configuration can be defined as 27 electrons distribution in 4 shells of Co element. The electron configuration of the Cobalt can be represented as the Ar 3d7 4s2 in its most accurate and precise form. Learn how to write the complete electron configuration of cobalt in two ways: through orbitals (Bohr model) and through sub-energy levels (Aufbau model).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
